基因組DNA Isolation Dual Kit (Column Based) | Bio-Helix 貨號PDC02-0100
Bio-Helix-貨號PDR04-0100-2.jpg)
| 代理廠牌: |  |
| 原廠連結: | |
| 相關下載: |
基因組Genomic DNA Isolation Dual Kit (Column Based)| Bio-Helix 貨號PDC02-0100
Genomic DNA Isolation Dual Kit (Column Based)
產品描述:
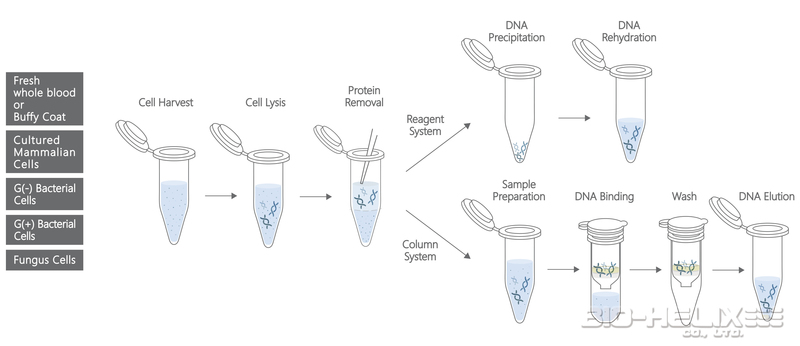
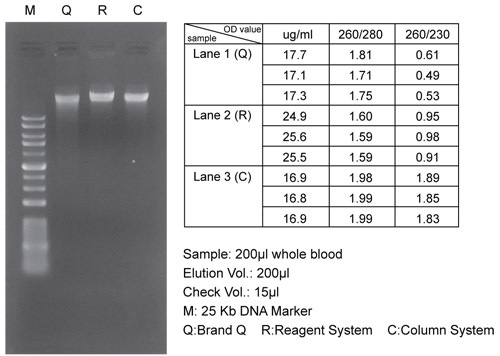
DUAL 基因組 DNA 分離試劑盒(血液/培養細胞/真菌)結合了試劑系統( reagent system)和離心柱系統(spin column system)。該試劑盒專為從全血( whole blood)、冷凍血液(frozen blood)、血沉棕黃層(buffy coat)、培養的動物/細菌細胞和真菌中分離基因組 DNA 而設計。這種獨特的試劑系統確保了從樣品中獲得的總 DNA( total DNA) 產量高、質量好。離心柱系統設計用於純化或濃縮先前已用試劑分離的 DNA 產物。無需苯酚/氯仿萃取( phenol/ chloroform extraction),整個過程可在 1 小時內完成。純化的 DNA 適用於 PCR 或其他酶促反應。
Sample
Up to 300 μl of the whole blood
Up to 200 μl of the frozen blood
Up to 200 μl of the buffy coat
Cultured animal cells (up to 1 x10^7)
Cultured bacterial cells (up to 1 x10^9)
Fungus cells (up to 5 x 10^7)
Format
Reagent and spin column
Yield
Up to 50 μg
Operation time
Within 60 minutes
Elution volume
50∼200 μl
▍Fresh Whole Blood or Buffy Coat
Reagent System Protocol
Step 1 – Sample Cells Harvesting
1. Collect blood in the EDTA-Na2 treated collection tubes (or other anticoagulant mixtures).
2. Transfer up to 300 µl of the blood or 200 µl of buffy coat to a sterile 1.5 ml microcentrifuge tube.
3. Add 900 µl of the Buffer RL and mix by inversion.
4. Incubate the tube at the room temperature for 10 minutes (invert twice during incubation).
5. Centrifuge at 4,000 x g for 5 minutes.
6. Remove the supernatant completely and resuspend the cells in 50 µl of the Buffer RL by pipetting the pellet.
Step 2 – Lysis
1. Add 300 µl of the Buffer CL to the resuspended cells from Step 1 and mix by vortex.
2. Incubate at 60°C for 10 minutes or until the sample lysate is clear. During the incubation, invert the tube every 3 minutes.
Optional Step: RNA Degradation (If RNA-free genomic DNA is required, perform this optional step.)
3. Add 5 µl of RNase A (10 mg/ml) to the sample lysate and mix by vortex. Incubate at room temperature for 5 minutes.
Step 3 – Protein Removal
1. Add 100 µl of the Buffer PO to the sample lysate and vortex immediately for 10 seconds.
2. Incubate on ice for 5 minutes.
3. Centrifuge at 14-16,000 x g for 3 minutes.
4. Transfer the supernatant to a clean 1.5 ml microcentrifuge tube.
Switch Step
◆ If more pure DNA is required, please switch to Column System (DNA Pure) Protocol.
Step 4 – DNA Precipitation
1. Add 300 µl of Isopropanol to the sample from the Step 3 and mix well by inverting 20 times.
2. Centrifuge at 14-16,000 x g for 5 minutes.
3. Discard the supernatant and add 300 µl of 70% ethanol to wash the pellet.
4. Centrifuge at 14-16,000 x g for 3 minutes.
5. Discard the supernatant and air-dry the pellet for 10 minutes.
Step 5 DNA Rehydration
1. Add 50-100 µl of the Buffer E and incubate at 60°C for 5-10 minutes to dissolve the DNA pellet. During the incubation, tap the bottom of the tube to promote DNA rehydration.
Column System (DNA Pure) Protocol
* Add 60ml of the absolute ethanol to the Buffer W2 prior to initial use.
* Pre-heat the Buffer E to 60°C prior to use.
Step 1 – Sample Preparation
1. Add 400 µl of the Buffer BD to the sample from Step 3 Protein Removal and shake vigorously.
Step 2 – DNA Binding
1. Place a DG Column in a 2 ml Collection Tube.
2. Transfer the sample mixture from the previous step to the DG Column.
3. Centrifuge at 14-16,000 x g for 30 seconds.
4. Discard the flow-through and place the DG Column back in the same Collection Tube.
Step 3 – Wash
1. Add 400 µl of the Buffer W1 into the DG Column.
2. Centrifuge at 14,000 x g for 30 seconds.
3. Discard the flow-through and place the DG Column back into the same Collection tube.
4. Add 600 µl of the Buffer W2 (Ethanol added) into the DG Column.
5. Centrifuge at 14,000 x g for 30 seconds.
6. Discard the flow-through and place the DG Column back into the same Collection tube.
7. Centrifuge at 14,000 x g again for 2 minutes to remove residual Buffer W2.
Step 4 – DNA Elution
1. Place the dried DG column in a clean 1.5 ml microcentrifuge tube.
2. Add 50-200 µl of Pre-Heated Buffer E or TE (not provided) into the center of the column matrix.
3. Let it stand at 60°C for 5 minutes.
4. Centrifuge for 2 minutes at 14-16,000 x g to elute the purified DNA.
▍Cultured Mammalian Cells
Reagent System Protocol
Step 1 – Sample Cells Harvesting
1. Transfer cultured mammalian cells (up to 10^7) to a sterile 1.5 ml microcentrifuge tube.
2. Centrifuge at 6,000 x g for 1 minute.
3. Remove the supernatant completely and resuspend the cells in 50 µl of the Buffer RL by pipetting the pellet.
Step 2 – Lysis
1. Add 300 µl of the Buffer CL to the resuspended cells from Step 1 and mix by vortex.
2. Incubate at 60°C for 10 minutes or until the sample lysate is clear. During the incubation, invert the tube every 3 minutes.
Optional Step: RNA Degradation (If RNA-free genomic DNA is required, perform this optional step.)
3. Add 5 µl of RNase A (10 mg/ml) to the sample lysate and mix by vortex. Incubate at room temperature for 5 minutes.
Step 3 – Protein Removal
1. Add 100 µl of the Buffer PO to the sample lysate and vortex immediately for 10 seconds.
2. Incubate on ice for 5 minutes.
3. Centrifuge at 14-16,000 x g for 3 minutes.
4. Transfer the supernatant to a clean 1.5 ml microcentrifuge tube.
Switch Step
◆If more pure DNA is required, please switch to Column System (DNA Pure) Protocol.
Step 4 – DNA Precipitation
1. Add 300 µl of Isopropanol to the sample from the Step 3 and mix well by inverting 20 times.
2. Centrifuge at 14-16,000 x g for 5 minutes.
3. Discard the supernatant and add 300 µl of 70% ethanol to wash the pellet.
4. Centrifuge at 14-16,000 x g for 3 minutes.
5. Discard the supernatant and air-dry the pellet for 10 minutes.
Step 5 – DNA Rehydration
1. Add 50-100 µl of the Buffer E and incubate at 60°C for 5-10 minutes to dissolve the DNA
pellet. During the incubation, tap the bottom of the tube to promote DNA rehydration.
Column System (DNA Pure) Protocol
* Add 60ml of the absolute ethanol to the Buffer W2 prior to initial use.
* Pre-heat the Buffer E to 60°C prior to use.
Step 1 – Sample Preparation
1. Add 400 µl of the Buffer BD to the sample from Step 3 Protein Removal and shake vigorously.
Step 2 – DNA Binding
1. Place a DG Column in a 2 ml Collection Tube.
2. Transfer the sample mixture from the previous step to the DG Column.
3. Centrifuge at 14-16,000 x g for 30 seconds.
4. Discard the flow-through and place the DG Column back in the same Collection Tube.
Step 3 – Wash
1. Add 400 µl of the Buffer W1 into the DG Column.
2. Centrifuge at 14,000 x g for 30 seconds.
3. Discard the flow-through and place the DG Column back into the same Collection tube.
4. Add 600 µl of the Buffer W2 (Ethanol added) into the DG Column.
5. Centrifuge at 14,000 x g for 30 seconds.
6. Discard the flow-through and place the DG Column back into the same Collection tube.
7. Centrifuge at 14,000 x g again for 2 minutes to remove residual Buffer W2.
Step 4 – DNA Elution
1. Place the dried DG column in a clean 1.5 ml microcentrifuge tube.
2. Add 50-200 µl of Pre-Heated Buffer E or TE (not provided) into the center of the column matrix.
3. Let it stand at 60°C for 5 minutes.
4. Centrifuge for 2 minutes at 14-16,000 x g to elute the purified DNA.
▍Gram-Negative Bacterial Cells
Reagent System Protocol
Step 1 – Sample Cells Harvesting
1. Transfer cultured bacterial cells (up to 10^9) to a sterile 1.5 ml microcentrifuge tube.
2. Centrifuge at 12,000 x g for 1 minute.
3. Remove the supernatant completely and resuspend the cells in 50 µl of the Buffer RL by pipetting the pellet.
Step 2 – Lysis
1. Add 300 µl of the Buffer CL to the resuspended cells from Step 1 and mix by vortex.
2. Incubate at 60°C for 10 minutes or until the sample lysate is clear. During the incubation, invert the tube every 3 minutes.
Optional Step: RNA Degradation (If RNA-free genomic DNA is required, perform this optional step.)
3. Add 5 µl of RNase A (10 mg/ml) to the sample lysate and mix by vortex. Incubate at room temperature for 5 minutes.
Step 3 – Protein Removal
1. Add 100 µl of the Buffer PO to the sample lysate and vortex immediately for 10 seconds.
2. Incubate on ice for 5 minutes.
3. Centrifuge at 14-16,000 x g for 3 minutes.
4. Transfer the supernatant to a clean 1.5 ml microcentrifuge tube.
Switch Step
◆If more pure DNA is required, please switch to Column System (DNA Pure) Protocol.
Step 4 – DNA Precipitation
1. Add 300 µl of Isopropanol to the sample from the Step 3 and mix well by inverting 20 times.
2. Centrifuge at 14-16,000 x g for 5 minutes.
3. Discard the supernatant and add 300 µl of 70% ethanol to wash the pellet.
4. Centrifuge at 14-16,000 x g for 3 minutes.
5. Discard the supernatant and air-dry the pellet for 10 minutes.
Step 5 – DNA Rehydration
1. Add 50-100 µl of the Buffer E and incubate at 60°C for 5-10 minutes to dissolve the DNA pellet. During the incubation, tap the bottom of the tube to promote DNA rehydration.
Column System (DNA Pure) Protocol
* Add 60ml of the absolute ethanol to the Buffer W2 prior to initial use.
* Pre-heat the Buffer E to 60°C prior to use.
Step 1 – Sample Preparation
1. Add 400 µl of the Buffer BD to the sample from Step 3 Protein Removal and shake vigorously.
Step 2 – DNA Binding
1. Place a DG Column in a 2 ml Collection Tube.
2. Transfer the sample mixture from the previous step to the DG Column.
3. Centrifuge at 14-16,000 x g for 30 seconds.
4. Discard the flow-through and place the DG Column back in the 2 ml Collection Tube.
Step 3 – Wash
1. Add 400 µl of the Buffer W1 into the DG Column.
2. Centrifuge at 14,000 x g for 30 seconds.
3. Discard the flow-through and place the DG Column back into the same Collection tube.
4. Add 600 µl of the Buffer W2 (Ethanol added) into the DG Column.
5. Centrifuge at 14,000 x g for 30 seconds.
6. Discard the flow-through and place the DG Column back into the same Collection tube.
7. Centrifuge at 14,000 x g again for 2 minutes to remove residual Buffer W2.
Step 4 – DNA Elution
1. Place the dried DG column in a clean 1.5 ml microcentrifuge tube.
2. Add 50-200 µl of Pre-Heated Buffer E or TE (not provided) into the center of the column matrix.
3. Let it stand at 60°C for 5 minutes.
4. Centrifuge for 2 minutes at 14-16,000 x g to elute the purified DNA.
▍Gram-Postive Bacterial Cells
Reagent System Protocol
Step 1 – Sample Cells Harvesting
1. Transfer cultured bacterial cells (up to 10^9) to a sterile 1.5 ml microcentrifuge tube.
2. Centrifuge at 12,000 x g for 1 minute.
3. Remove the supernatant completely and resuspend the cells in 100 µl of lysozyme Buffer by pipetting the pellet.
4. Incubate at room temperature for 20 minutes.
Step 2 – Lysis
1. Add 300 µl of the Buffer CL to the resuspended cells from Step 1 and mix by vortex.
2. Incubate at 60°C for 10 minutes or until the sample lysate is clear. During the incubation, invert the tube every 3 minutes.
Optional Step: RNA Degradation (If RNA-free genomic DNA is required, perform this optional step.)
3. Add 5 µl of RNase A (10 mg/ml) to the sample lysate and mix by vortex. Incubate at room temperature for 5 minutes.
Step 3 – Protein Removal
1. Add 100 µl of the Buffer PO to the sample lysate and vortex immediately for 10 seconds.
2. Incubate on ice for 5 minutes.
3. Centrifuge at 14-16,000 x g for 3 minutes.
4. Transfer the supernatant to a clean 1.5 ml microcentrifuge tube.
Switch Step
◆If more pure DNA is required, please switch to Column System (DNA Pure) Protocol.
Step 4 – DNA Precipitation
1. Add 300 µl of Isopropanol to the sample from the Step 3 and mix well by inverting 20 times.
2. Centrifuge at 14-16,000 x g for 5 minutes.
3. Discard the supernatant and add 300 µl of 70% ethanol to wash the pellet.
4. Centrifuge at 14-16,000 x g for 3 minutes.
5. Discard the supernatant and air-dry the pellet for 10 minutes.
Step 5 – DNA Rehydration
1. Add 50-100 µl of the Buffer E and incubate at 60°C for 5-10 minutes to dissolve the DNA pellet. During the incubation, tap the bottom of the tube to promote DNA rehydration.
Column System (DNA Pure) Protocol
* Add 60ml of the absolute ethanol to the Buffer W2 prior to initial use.
* Pre-heat the Buffer E to 60°C prior to use.
Step 1 – Sample Preparation
1. Add 400 µl of the Buffer BD to the sample from Step 3 Protein Removal and shake vigorously.
Step 2 – DNA Binding
1. Place a DG Column in a 2 ml Collection Tube.
2. Transfer the sample mixture from the previous step to the DG Column.
3. Centrifuge at 14-16,000 x g for 30 seconds.
4. Discard the flow-through and place the DG Column back in the 2 ml Collection Tube.
Step 3 – Wash
1. Add 400 µl of the Buffer W1 into the DG Column.
2. Centrifuge at 14,000 x g for 30 seconds.
3. Discard the flow-through and place the DG Column back into the same Collection tube.
4. Add 600 µl of the Buffer W2 (Ethanol added) into the DG Column.
5. Centrifuge at 14,000 x g for 30 seconds.
6. Discard the flow-through and place the DG Column back into the same Collection tube.
7. Centrifuge at 14,000 x g again for 2 minutes to remove residual Buffer W2.
Step 4 – DNA Elution
1. Place the dried DG column in a clean 1.5 ml microcentrifuge tube.
2. Add 50-200 µl of Pre-Heated Buffer E or TE (not provided) into the center of the column matrix.
3. Let it stand at 60°C for 5 minutes.
4. Centrifuge for 2 minutes at 14-16,000 x g to elute the purified DNA.
▍Fungus Cells
Reagent System Protocol
Step 1 – Sample Cells Harvesting
1. Transfer fungus cells (up to 10^8) to a sterile 1.5 ml microcentrifuge tube.
2. Centrifuge at 6,000 x g for 5 minutes.
3. Remove the supernatant completely and resuspend the cells in 600 µl of sorbitol Buffer by pipetting the pellet.
4. Add 200 U of lyticase or zymolase. Incubate at 30°C for 30 minutes.
5. Centrifuge the mixture for 10 minutes at 2,000 x g to harvest the spheroplast.
6. Remove the supernatant completely and resuspend the cells in 50 µl of the Buffer RL by pipetting the pellet.
Step 2 – Lysis
1. Add 300 µl of the Buffer CL to the resuspended cells from Step 1 and mix by vortex.
2. Incubate at 60°C for 10 minutes or until the sample lysate is clear. During the incubation, invert the tube every 3 minutes.
Optional Step: RNA Degradation (If RNA-free genomic DNA is required, perform this optional step.)
3. Add 5 µl of RNase A (10 mg/ml) to the sample lysate and mix by vortex. Incubate at room temperature for 5 minutes.
Step 3 – Protein Removal
1. Add 100 µl of the Buffer PO to the sample lysate and vortex immediately for 10 seconds.
2. Incubate on ice for 5 minutes.
3. Centrifuge at 14-16,000 x g for 3 minutes.
4. Transfer the supernatant to a clean 1.5 ml microcentrifuge tube.
Switch Step
◆If more pure DNA is required, please switch to Column System (DNA Pure) Protocol.
Step 4 – DNA Precipitation
1. Add 300 µl of Isopropanol to the sample from the Step 3 and mix well by inverting 20 times.
2. Centrifuge at 14-16,000 x g for 5 minutes.
3. Discard the supernatant and add 300 µl of 70% ethanol to wash the pellet.
4. Centrifuge at 14-16,000 x g for 3 minutes.
5. Discard the supernatant and air-dry the pellet for 10 minutes.
Step 5 – DNA Rehydration
1. Add 50-100 µl of the Buffer E and incubate at 60°C for 5-10 minutes to dissolve the DNA pellet. During the incubation, tap the bottom of the tube to promote DNA rehydration.
Column System (DNA Pure) Protocol
* Add 60ml of the absolute ethanol to the Buffer W2 prior to initial use.
* Pre-heat the Buffer E to 60°C prior to use.
Step 1 – Sample Preparation
1. Add 400 µl of the Buffer BD to the sample from Step 3 Protein Removal and shake vigorously.
Step 2 – DNA Binding
1. Place a DG Column in a 2 ml Collection Tube.
2. Transfer the sample mixture from the previous step to the DG Column.
3. Centrifuge at 14-16,000 x g for 30 seconds.
4. Discard the flow-through and place the DG Column back in the 2 ml Collection Tube.
Step 3 – Wash
1. Add 400 µl of the Buffer W1 into the DG Column.
2. Centrifuge at 14,000 x g for 30 seconds.
3. Discard the flow-through and place the DG Column back into the same Collection tube.
4. Add 600 µl of the Buffer W2 (Ethanol added) into the DG Column.
5. Centrifuge at 14,000 x g for 30 seconds.
6. Discard the flow-through and place the DG Column back into the same Collection tube.
7. Centrifuge at 14,000 x g again for 2 minutes to remove residual Buffer W2.
Step 4 – DNA Elution
1. Place the dried DG column in a clean 1.5 ml microcentrifuge tube.
2. Add 50-200 µl of Pre-Heated Buffer E or TE (not provided) into the center of the column matrix.
3. Let it stand at 60°C for 5 minutes.
4. Centrifuge for 2 minutes at 14-16,000 x g to elute the purified DNA.

PCR混合液 | 100bp-10kb DNA Ladder | 瓊脂膠體粉Agarose | DNA核酸染劑 | 藍光觀察箱(含白光) | Mupid-2 水平電泳槽 |

![]()
bio-helix.com
Bio-Helix成立於2007年,專注於為全球生命科學研究人員提供最好的生物試劑。包括 DNA 標記物、蛋白質標記物、預製蛋白質凝膠、核酸純化、PCR試劑等。與世界各地實驗室的科學家們一起努力,不斷提高質量,開始贏得客戶的信任和評價。品牌遍布四十多個國家,已成為生命科學實驗室值得信賴和歡迎的試劑品牌。Bio-Helix個願景,就是有一天我們的壽命可以延長;所有疾病的早期發現和隨後的適當治療都可以提高生活質量。中國古人認為,良醫治未病,治未病。在 Bio-Helix 中,創建了一個新品牌 LifeDirex 來實現這一願景。 LifeDirex 將專注於為診斷工具提供最好和最具創新性的試劑,以促進盡可能早地發現疾病。Bio-Helix使命是提供最優質的產品並開發新技術,幫助研究人員找到尋求科學成就的解決方案。此外,Bio-Helix認為現在是進入體外診斷設備(IVDD)市場的最佳時機。我們的目標是成為研究實驗室和IVDD的最佳解決方案提供商。
在改善生活質量和實現科學卓越的願望的驅使下,Bio-Helix以最高的誠信標準運營。還將這種精神延伸到我們的服務和全球價值中,以提供給世界各地的客戶。多樣化的員工隊伍和協作網絡形成了公司文化的完美結合。 Bio-Helix 不僅是一家機會均等的公司,而且還努力提供友好和成長的環境。
| CAT# | NAME | Size |
| BP001CU | BluPAD, LED Transilluminator, with mini Darkroom (CE/ETL, Patent No: M543442) |
Set |
| PMI09-0500 | IRIS9 Plus Prestained Protein Ladder (9 pre-stained bands, 15-170 kDa) |
500 μl |
| PMI11-0500 | IRIS11 Prestained Protein Ladder (11 pre-stained bands, 3-260 kDa) |
500 μl |
| PMB01-0500 | Blu10 Plus Prestained Protein Ladder (10 pre-stained bands, 6.5-270 kDa) |
500 μl |
| PMB10-0500 | Blu10 Prestained Protein Ladder (10 pre-stained bands, 11-180 kDa) |
500 μl |
| PMB11-0500 | Blu11 Prestained Protein Ladder (11 pre-stained bands, 10-180 kDa) |
500 μl |
| PMB12-0500 | Blu12 Prestained Protein Ladder (12 pre-stained bands, 11-245 kDa) |
500 μl |
| PMB13-0500 | Blu13 Prestained Protein Ladder (13 pre-stained bands, 5-245 kDa) |
500 μl |
| PMU12-0500 | UNveil Unstained Protein Ladder, Broad Range (12 unstained bands, 10-200 kDa) |
500 μl |
| CCH321-B100ML | UltraScence Pico Plus Western Substrate (Patent No: US10,711,185) |
100 ml (50ml+50ml) |
| CCH345-B100ML | UltraScence Pico Ultra Western Substrate (Patent No: US10,711,185) |
100 ml (50ml+50ml) |
| CCH365-B100ML | UltraScence Femto Western Substrate (Patent No: US10,711,185) |
100 ml (50ml+50ml) |
| CCH375-B100ML | UltraScence Femto Plus Western Substrate (Patent No: US10,711,185) |
100 ml (50ml+50ml) |
| PS002-B500ML | COOMASSIEnano Protein Staining Solution, 500ml | 500 ml |
| PS003-B500ML | Ponceau S Protein Staining Solution | 500 ml |
| BS001-B500ML | OneStep Blocker Western Blocking Solution and Signal Enhancer |
500 ml |
| TM001-B200ML | TMB ELISA Substrate | 100 ml (50ml+50ml) |
| DM001-R500 | BH 100bp DNA Ladder RTU (11 bands, 100-1,500bps) |
500 μl |
| DM003-R500 | BH 100bp DNA Ladder H3 RTU (12 bands, 100-3,000bps) |
500 μl |
| DM012-R500 | BH 50bp DNA Ladder RTU (17 bands, 50-1,500bps) |
500 μl |
| DM015-R500 | BH 1Kb Plus DNA Ladder RTU (13 bands, 100-10,000bps) |
500 μl |
| DMF12-0100 | OmniMARK 100 DNA Ladder RTU (12 bands, 100-3,000bps, fluorescent dye) |
600 μl |
| DMF13-0100 | OmniMARK 1K DNA Ladder RTU (13 bands, 100-10,000bps, fluorescent dye) |
600 μl |
| LD011-1000 | Prime Juice Preloading Fluorescent Stain | 1 ml |
| LD003-0500 | Novel Green Plus (20,000X) DNA Staining Reagent | 500 μl |
| AGT002-0500 | Agarose Tablets, 110 pcs | 0.5g x 110 |
| MB755-0100 | Agarose Powder 100g -Molecular Biology Grade | 100 g |
| MB755-0500 | Agarose Powder 500g -Molecular Biology Grade | 500 g |
| MB101-0500 | Taq DNA Polymerase | 500U/100μl |
| DP001-0100 | nanoTaq™ Hot-Start DNA Polymerase | 100 rxns 500U/100μl |
| DN001-0250 | 100 mM dNTP Set | 4 x 250μl |
| DN0010 | 10 mM dNTP Mix | 1 ml |
| MB200-P100 | 2X PCR SuperMix | 100 rxns 2 x 1.25mL |
| MBA01-0100 | OmniPCR Supermix w Fluorescent dye | 100 rxns (2.5ml) |
| RK001-0050 | RScript™ cDNA Synthesis Kit | 50 rxns |
| RT001-0050 | RScript™ Reverse Transcriptase 10,000U | 50 rxns 10,000U/50μl |
| RT001-0250 | RScript™ Reverse Transcriptase 50,000U | 250 rxns 50,000U/250μl |
| RI001-0125 | RIBOAssure™ RNase Inhibitor 5,000U | 125 rxns 5,000U/125μl |
| QPR01-0100 | PanProbes™ One-Step RT-qPCR Kit | 100 reactions (20 μl vol) |
| QPD01-0100 | PanProbes™ Universal qPCR MasterMix | 100 reactions (20 μl vol) |
| QSR01-0100 | PanGreen™ One-Step RT-qPCR Kit | 100 reactions (20 μl vol) |
| QSD01-0100 | PanGreen™ Universal SYBR® Green Mastermix | 100 reactions (20 μl vol) |
| PDP01-0100 | Plasmid miniPREP Kit | 100 rxns |
| PDP02-0020 | Plasmid midiPREP Kit | 20 rxns |
| PDP03-0010 | Plasmid maxiPREP Kit | 10 rxns |
| PDC01-0100 | PCR Clean-Up & Gel Extraction Kit | 100 rxns |
| PDC02-0100 | Genomic DNA isolation dual kit | 100 rxns |
| PDC05-0100 | Dual Genomic DNA Isolation Kit (Plant) | 100 rxns |
| PDC06-0100 | Dual Genomic DNA Isolation Kit(Tissue) | 100 rxns |
| PDC09-0100 | Genomic DNA Isolation Kit (Blood/Cultured Cell/Fungus) | 100 rxns |
| PDC10-0100 | Genomic DNA Isolation Kit (Plant) | 100 rxns |
| PDC11-0100 | Genomic DNA Isolation Kit (Tissue) | 100 rxns |
| PDC12-0100 | Genomic DNA Isolation Kit (Paraffin-embedded tissue) | 100 rxns |
| PDM01-0100 | Mbead Buffy Coat Genomic DNA Kit | 100 rxns |
| PDM02-0100 | Mbead Tissue Genomic DNA Kit | 100 rxns |
| PDM03-0100 | Mbead Bacteria Genomic DNA Kit | 100 rxns |
| PDM05-0100 | Mbead Plant Genomic DNA Kit | 100 rxns |
| PDM06-0100 | Mbead Blood/Cell Genomic DNA Kit | 100 rxns |
| PDR01-0100 | Genomic DNA Isolation Reagent | 100 rxns |
| PDR02-0100 | Plant Genomic DNA Isolation Reagent | 100 rxns |
| PDR05-0100 | Genomic DNA Isolation Reagent Kit(Blood/Cultured Cell/Tissue) | 100 rxns |
| PDC04-0100 | Total RNA Isolation Kit (Blood/Cultured Cell/Fungus) | 100 rxns |
| PDC07-0100 | Total RNA Isolation Kit (Plant) | 100 rxns |
| PDC08-0100 | Total RNA Isolation Kit(Tissue) | 100 rxns |
| PDR03-0100 | Total RNA Isolation Reagent | 100 rxns |
| PDR04-0100 | Plant Total RNA Isolation Reagent | 100 rxns |
| PDC03-0100 | Virus Nucleic Acid Isolation Kit | 100 rxns |
| PDM04-0100 | Mbead Virus Genomic Nucleic Acid Kit | 100 rxns |
| QP019M-0050 | COVID-19 Mutation RT-qPCR Detection Kit (Alpha, Delta, Kappa) | 50 rxns |
| QP019T-0100 | COVID-19 RT-qPCR Detection Kit Plus (TFDA) | 100 rxns |
| QP019-0100 | SARS-CoV-2/COVID-19 RT-qPCR Detection Kit (RUO) | 100 rxns |